Abid Latif Qureshi ( KRL Hospital, Islamabad. )
Kauser Abid ( Islamabad Medical & Dental College, Islamabad. )
August 2010, Volume 60, Issue 8
Original Article
Abstract
Objective: To investigate the incidence of bacteraemia and bacterial colonization related to the use of dual-lumen catheters in ESRD patients on haemodialysis.
Methods: Sixty patients with ESRD of varied etiologies, both males and females falling in the age range (16-74 years) were randomly selected. Non-cuffed, non-tunneled polyurethane double lumen catheters were inserted under aseptic technique. Patients on twice a week schedule of haemodialysis were followed up for a period of 5 months. After every haemodialysis session, catheters were examined for any local infection or signs of bacteraemia. In case of suspicion, distal 5 cm segment of the catheter, and local pus swab and two blood culture samples were sent to the pathology laboratory.
Result: Thirty one catheters (51.6%) sent for culture and sensitivity showed colonization (>15 CFU). Bacteraemia was positive in 15 (25%) patients. Thirteen (41.9%) catheter tips were found to be colonized by staphylococcus epidermidis, eleven (35.4%) by staphylococcus aureus, three (0.96%) by Candida albicans species, two (6.45%) by E coli, one (3.2%) by P Aeruginosa and one (3.2%) by mixed Pseudomonas and E coli respectively.
Conclusion: Non cuffed non tunneled double lumen catheters are designed for short term emergency use and should be used in the same context. Although pathogenesis of catheter related infection is multifactorial the transcutaneous migration of organisms colonizing the skin remains the most important route (JPMA 60:671; 2010).
Introduction
Prior to the availability of renal replacement modalities, little hope was available for patients afflicted with end stage renal disease (ESRD). In the early 1960\'s, the advent of haemodialysis revolutionized the treatment of renal failure and contributed significantly in sustaining the lives of patients suffering from irreversible loss of renal function.1 However, haemodialysis is not exempt from complications, notably those which are vascular access related.
Vascular access for haemodialysis could be temporary or permanent. Permanent vascular access is by surgical creation of an arteriovenous fistula or implantation of arterio venous graft.2 Following fistula creation, an average of 6 weeks is required for fistula maturation. Its premature use for haemodialysis can result in permanent loss of fistula. In case of arteriovenous graft, two weeks are required for healing of subcutaneous tunnel. So temporary vascular access is helpful in cases where permanent vascular access is either not available or not ready to be used. It is also useful in patients with reversible form of acute renal failure where permanent vascular access is not justified. In temporary vascular access, percutaneous venous cannula is placed into large intra thoracic veins via subclavian or jugular route or into femoral vein. They are easy to be placed by experienced hands, can be used immediately after placement for haemodialysis and provide excellent extra corporeal blood flow rates.3
Organisms can gain entry to an intra vascular access at several points. These include direct entry of micro organisms at the catheter insertion site during placement, secondary contamination of infusate and seeding of the catheter by micro organisms during bacteraemia. The skin normally harbours 103-104 micro organisms per cm2 of skin. Most of these are commensals like Staphylococcus Epidermidis, Staph. Aureus, Corynebacterium, various Streptococci, Pseudomonas Aerugenosa, anaerobes like Peptococcus, and yeasts like Candida albicans.
Kozney et al in 19844 evaluated 74 nontunelled subclavian catheters and documented Staph. aureus as the most commonly isolated organism. Cheesbrough et al5 evaluated 64 subclavian haemodialysis catheters and likewise found Staph. Aureus as major microorganism responsible for infection. Almirall et al6 and Kirkpatrick et al7 in their studies revealed the same results.
If adequate aseptic measures are not observed during catheter insertion (hand wash, gown, mask and gloves worn by the doctor), the organisms can readily gain entry into insertion site and start replication. This study was planned to evaluate the prevalence of bacterial colonization and the causative organisms isolated from dual-lumen catheters in patients on haemodialysis.
Patients and Methods
It was a prospective observational study of descriptive nature. The study was conducted at the Nephrology department of Pakistan Institute of Medical Sciences Islamabad. Overall 60 patients undergoing haemodialysis were selected randomly not considering age or gender matching. The study spanned over five months with the inclusion criteria being patients with chronic renal failure of varying etiology in whom renal replacement therapy in the form of haemodialysis was needed and a definite vascular access (an arterio-venous fistula) was not available or mature at the time of the study. Exclusion criteria were patients with acute renal failure (because haemodialysis is usually required for a short duration in these cases), patients with chronic renal failure having a functioning AV fistula, patients with already established septicaemia as evidenced by fever, leukocytosis or septic shock, and in cases where there was an accidental breach in the aseptic precautions during catheter insertion.
Non cuffed poly-urethane catheters were used {Med Comp(R)8 and Arrow international (R)}. catheters were inserted under aseptic technique in the procedure room of the Haemodialysis unit. In 24 patients , the femoral route was used, 31 catheters were inserted into the subclavian vein using the infraclavicular approach described by Uldell et al.3 In the remaining 5 patients catheters were inserted into the internal jugular vein using the central approach. The catheters were used only for haemodialysis and intradialytic infusions.
At each visit of the patient for haemodialysis, a subjective enquiry was made regarding the presence of fever, local discomfort or discharge. Every patient was followed up till catheter was removed for reasons like infection, catheter blockage due to intraluminal clotting or a kink, and limb swelling due to venous thrombosis.
Catheter exit-site infection was defined as erythema, tenderness and pus at the catheter insertion site and unaccompanied by fever.
Severe infection (Catheter-related bacteraemia) was defined as patients presenting with high grade fever or septic shock with a temporary haemodialysis catheter in place and no alternative source of infection could be documented. Exit-site infection may or may not have been present.
Criteria for line removal was any of the following:
Severe infection, Catheter blockage, Venous thrombosis due to presence of catheter, Significant patient\'s discomfort due to catheter, AV fistula maturation (catheter no longer needed), and patient\'s death.
In case of local catheter related infection skin swabs were taken and sent to the laboratory along with two blood samples (one each from central venous catheter and from peripheral vein) for culture, as 10 ml blood sample added to 100 ml of growth medium brain heart broth infusion. In case of severe infection requiring catheter removal, the catheter was flushed with 10 ml of 0.9% saline, skin cleaned with antiseptic agent (Povidone-Iodine) and allowed to dry. Catheters were clamped with sterile forceps as close to skin as possible, sectioned distally with sterile blade and carefully removed avoiding skin contact. They were immediately placed in sterile container and sent to the laboratory for semi quantitative analysis.
Laboratory processing: Skin swabs were plated onto blood agar and incubated aerobically for 48-72 hrs. Any isolate present with more then 5 CFU was regarded as significant for exit site infection.
Blood cultures: Samples were incubated aerobically at 37°C for 7 days. They were subcultured after 7 days (turbidity found) onto blood agar and incubated aerobically at 37°C. The organisms were speciated and typed. Antibiotic sensitivity was obtained by using lab protocols (by disk diffusion assay)
Laboratory protocol for Catheter cultures: (by semi quantitative culture method described by Dennis G Maki et al 1997)9 Catheter segments were rolled back and forth 4 times on a surface of 100 mm 5% sheep blood agar plate. Catheter segments showing bending or got bent were smeared. The segment was immersed in tryptase soy broth. Both plates and broths are incubated at 37°C. Turbid broth and broth cultures without apparent growth after 72 hours were subcultured onto blood agar plates which was processed similarly on primary plates, were enumerated and all organisms received from both primary plates and broth were fully identified
Interpretation:
Less then 15 colonies/plate = contamination, no catheter related sepsis
More then 500 colonies /plate= catheter related sepsis present
Between 15- 500 colonies/plate= exit site infection. Catheter related infection may or may not be present.
Quantitative culture method: In 1979 Denis J Cleri et al10 described a modification of technique by Seligman in 197411 for quantitative culture. In the protocol intra vascular catheter segments were immersed in 2-10 ml of tryptase soy broth. A needle was inserted into proximal end of intravascular segment and flushed 3 times. The broths were serially diluted 100 folds and 0.1 ml of each dilution streaked onto sheep blood agar plate and incubated. Undiluted broths were also incubated. Organisms were identified by standard methods and antibiotic susceptibility tested by disk diffusion method. Colonies were counted after incubation for 72 hours at 37°C.
The number of colony forming units (CFU) were calculated by counting the colonies and multiplying 10 times the dilution factor and dividing the volume of broth in which the insert had been immersed.
Intravascular insert was considered infected if it was more then 103 CFU.
Intravascular insert- related bacteraemia was defined as the isolation of an organism of the same species and sensitivity pattern from both intravascular insert and simultaneous peripheral blood culture.
By combining quantitative and semi quantitative culture techniques, both the external and internal surface of catheters could be cultured, thus increasing sensitivity for identification of organisms.12
Statistical analysis:
The results were compared using Chi square (x2) test with Yates correction or Fishers\'s exact test when necessary to calculate the probability of remaining free from catheter-related bacteraemia.
Results
On examination nineteen out of sixty patients (31.66%) showed signs of local inflammation (pain, erythema, tenderness and pus exudation) at catheter insertion site. Pus swabs taken from insertion site revealed Staph Aureus in 7 cases, Staph Epidermidis in 6 cases, Candida species in 03 cases and one case each was infected with E.coli and Pseudomonas aeruginosa. In the remaining one case the infection was polymicrobial (mixed E.coli and Pseudomonas).
The distal 5 segments of all the catheters were sent for culture and sensitivity upon removal. It was revealed that 31 catheters (51.6%) were colonized (> 15 CFU). The organisms found on catheter surface were as shown in the Table-1.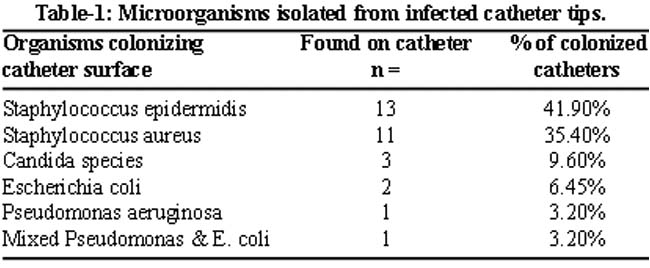
Bacteraemia was documented in 15 patients (25%). In four cases, catheter related bacteraemia was suspected on clinical grounds because of high grade fever with chills and rigors. In these patients there was no obvious source of infection to explain for bacteraemia and local inflammatory signs at catheter insertion site were unremarkable. Even milking at the catheter insertion site failed to reveal any pus exudation. The infecting organisms were Staph. Aureus in 2 cases and E. Coli and Staph Epidermidis in one case each. In 14 instances, catheter tip and blood cultures revealed the same organisms. In 12/15 patients, blood cultures drawn from line and periphery showed the same organisms. Blood cultures were negative in 45 patients (75%)
The characteristics of bacteraemia are as shown in Table-2.
To identify the possible factors associated with the development of catheter-related bacteraemia, a univariate analysis was done. Table-3 shows the results.
Discussion
The results show that infections frequently complicate double lumen catheters used for dialysis in our unit. Most of the western studies have evaluated the incidence of infections in cuffed, tunneled double lumen catheters or antibiotic-impregnated catheters which are not readily available and hence infrequently used in our local setting. Hence temporary non-cuffed non-tunneled double lumen catheters were evaluated for infection.
The results of our study were comparable in frequency as found by Almirall et al6 where they found a catheter colonization rate of 55% and bacteraemia in 17% patients. Lilyan et al,13 in a recent study documented 53.5% bacterial growth in temporary catheters with Staph. Epidermidis being the principal microorganism. Catheter colonization rate was 51.6% and bacteraemia was documented in 18% cases
Sanavi Suzan et al in 200714 observed Staph. aureus in 67% cases responsible for catheter related infection in their study group which is close to the figures revealed in our study. But they retained catheters for 30 days in 60 % of their patients which is longer than the recommended 3 weeks duration for non cuffed catheters.13,14
Regarding the source of bacterial colonization of noncuffed, non-tunneled catheters, considerable controversy exists. Most commonly, the skin around the catheter insertion site is implicated in allowing the ingress of microorganisms on the exterior surface of the catheter. Other research workers have mentioned catheter hub and contaminated infusate as causing catheter-related infection and bacteraemia.17 The present study was limited in finding the source of infection, since quantitative cultures of catheter hub and intraluminal portions were not performed. However the great preponderance of both catheter colonization and bacteraemia were caused by skin commensals (Staph aureus and Staph epidermidis) which indicates that transcutaneous migration of microorganisms may be a major cause of catheter colonization and subsequently, bacteraemia. This finding is supported by the fact that thirteen out of nineteen skin culture results (68%) revealed Staphylococci. In our study, however, four patients with bacteraemia did not exhibit any sign of local inflammation and they merely presented with malaise and high grade fever. These patients were found to have no other potential source of bacteraemia and double lumen catheter was removed only on clinical suspicion as the possible cause of bacteraemia. These patients promptly responded to catheter removal and antibiotics. This type of bacteraemia without any signs and symptoms of local catheter related infection was observed by Kozeny et al4 and our study confirmed its occurrence. We failed to find the source of bacteraemia but it was mentioned in a recent study by Kassaler et al5 that although the patients with CRF who are on chronic haemodialysis are more likely to develop bacteraemia from multiple foci (vascular access, urinary tract, biliary tract, skin and soft tissue), the source may be unknown in a substantial group of patients.
In 2002 Saeed Abdul Rahman et al18 compared the incidence of infection between temporary and permanent vascular access for haemodialysis. They found a higher infection rate in temporary vascular access but Staph epidermidis was isolated in 50% cases in their study group and similar results were documented by Pidro et al in 2007.19 Our study focused on temporary vascular access cases only and Staph. Aureus was responsible for infection in 50% of the cases, which is comparable to Tsu-yuan Chang\'s study reported in 2002.18
In the present study we found that the only factor associated with increased risk of catheter related infection was the duration of catheter insertion. This risk increases after 2 weeks of catheter insertion but the result was not statistically significant.
Conclusion
Infection frequently complicates double lumen catheters used for haemodialysis. The present study identified the duration of catheter insertion to be the only risk factor. This increases after 2 weeks of catheter insertion. Although double lumen catheters provide satisfactory means for haemodialysis, they are frequently complicated by infections. The non-cuffed, non tunneled catheters are designed for short term use and should be reserved for the same.
It was inferred that to minimize the occurrence of catheter-related infections, a double lumen catheter should be removed within two weeks (before infection is established) and be replaced by another catheter at an alternative site.
Acknowledgement
The assistance for the study provided by Mr. Haresh Kumar, Scientific Information officer, PASTIC National Center, Quaid-e-Azam University, Islamabad, is gratefully acknowledged.
The haemodialysis staff and Pathology laboratory staff of Pakistan Institute of Medical Sciences, Islamabad are thanked for their co-operation and help.
References
1.Banomini A, Feletti C, Scolari MP, Stefonis. Benefits of early initiation of dialysis. Kidney Int Suppl 1985; 17: 557-9.
2.Garabed E, Nathan L, Earl S. The National Kidney Foundation Dialysis Outcome Quality Initiative. Curr Opin Nephrol Hypertension 1997; 6.
3.Uldall PR, Woods F, Bird M, Dyck R. Subclavian cannula for temporary haemodialysis. Proc Clin Dial Transplant Forum 1979; 9: 268-72.
4.Gregory A. The carcinogenic potential of benzidine-based dyes. J Enviran Pathol Toxicol Oncol 1984; 5: 243-59.
5.Cheesbrough JS, Finch RG, Burden RP. A prospective study of the mechanism of Infection associated with haemodialysis catheters. J Infect Dis 1986; 154: 579-89.
6.Almirall J, Gonzalez J, Rello J, Campistol JM, Montoliu J, Puig de la Bellacasa J, et al. Infections of haemodialysis catheters: Incidence and mechanisms. Am J Nephrol 1989; 9: 454-9.
7.Kirkpatrick WG, Culpepper RM, Sirmon MD. Frequency of complications with prolonged femoral vein catheterization for haemodialysis access. Nephron 1996; 73: 58-62.
8.Lund G, Trerotola, Osterman FA. Translumbar inferior vena cava catheterization for haemodialysis access. J Am Soc Nephrol 1993; 4: 336.
9.Maki DG, Stolz SM, Wheeler S, Mermel LA. Prevention of central venous Catheter related blood stream infection by the use of an antiseptic impregnated catheter. A randomized controlled trial. Ann Intern Med 1997; 127: 257-66.
10.Cleri DJ, Corado ML, Seligman SJ. Quantitative culture of intravenous catheters and other intravenous inserts. J Infect Dis 1980; 141: 781-6.
11.Ryan JA Jr, Abel RM, Abbott WM, Hopkins CC, Chesney TM, Colley R et al. Catheter complications in total parenteral nutrition. A prospective study of 200 consecutive patients. N Eng J Med 1974; 290: 757-61.
12.Linares J, Stiges Serra A, Garau J, Perez JL, Martin R. Pathogenesis of catheter sepsis: a prospective study with quantitative and semi quantitative cultures of catheter hub and segment. J Clin Microbiol 1985; 21: 357-60.
13.de Freitas LW, Neto MM, Nascimento MM, Figuerredo JF. Bacterial colonization in haemodialysis temporary dual lumen catheters: a prospective study. Ren Fail 2008; 30: 31-5.
14.Sanavi S, Ghods A, Afshar R. Catheter associated infections in haemodialysis patients. Saudi J Kidney Dis Transpl 2007; 18: 43-6.
15.Dangerdos JI. Handbook of dialysis. 3rd ed., Philadelphia: Lippincott Williams & Wilkins 2001; 4: 43; 50, 28; 496-500
16.Miller DL, O\'Crady NP. Guidelines for prevention of intravascular catheter related infections: recommendations relevant to interventional radiology. J VAsc Inter Radiol 2003; 14: S355.
17.Salzman MB, Isenberg H, Shapiro JF, Lipsitz PJ, Rusin LG. A prospective study of catheter hub as the Portal of entry for microorganisms causing catheter-related sepsis in neonates. J Infect Dis 1993; 167: 487-90.
18.Rahman SA, Al-Mueilo SH, Bokhary HA, Ladipo GO, Al-Rubaish A. A prospective study of haemodialysis access related bacterial infections. J Infect Chemo-ther 2002; 8: 242-6.
19.Ponce P, Cruz J, Ferreira A, Oliveira C, Vinhas J, Silva G. A prospective study on incidence of bacterial infection in portugnese dialysis unit. Nephron Clin Pract 2007; 107: c133-8.
20.Chang TY, Fang HC, Tzeng HM, Chou KJ, Wu MJ, Tsai CC, et al. Infection associated with temporary central venous catheters for Haemodialysis. A review of 5 years experience. Acta Nephrologica 2002; 16: 12-8.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




