Kulsoom Farhat ( Department of Pharmacology & Therapeutics, Army Medical College, (National University of Sciences and Technology), Rawalpindi. )
Akbar Waheed ( Department of Pharmacology & Therapeutics, Army Medical College, (National University of Sciences and Technology), Rawalpindi. )
Azhar Hussain ( Hamdard Institute of Pharmaceutical Sciences, Islamabad. )
Muhammad Ismail ( Institute of Biomedical and Genetic Engineering, Islamabad. )
Qaisar Mansoor ( Institute of Biomedical and Genetic Engineering, Islamabad. )
Anwar Kamal Pasha ( Department of Anesthesia, Combined Military Hospital, Rawalpindi. )
Nusrat Jafery ( Department of Pharmacology & Therapeutics, Margalla Institute of Dentistry, Islamabad. )
September 2015, Volume 65, Issue 9
Original Article
Abstract
Objective: To study the association of C1236T single-nucleotide polymorphisms of ABCB1 gene with non-responsiveness to antiemetic treatment in post-operative patients.
Methods: The prospective, clinical trial was conducted at Combined Military Hospital, Rawalpindi, and the Institute of Biomedical and Genetic Engineering, Islamabad, in 2012-13, and comprised patients undergoing elective laparoscopic cholecystectomy. All patients were given 0.1 mg/kg ondansetron intravenously 30 minutes before the end of surgery and Deoxyribonucleic acid samples were obtained. The frequencies of genotypes of Single Nucleotide Polymorphism were determined by polymerase chain reaction followed by restriction fragment length polymorphism.
Results: Of the 426 patients, 201(47%) were responders having no nausea or vomiting, and 225(52.8%) were non-responders having nausea or vomiting. The incidence of post-operative nausea and vomiting during the first 2 hours after surgery was significantly lower in patients with 1236TT genotype than other 1236 genotypes (p<0.001). It was significantly higher in patients with CC genotype at 2 hours than other 1236 genotypes (p<0.001).
Conclusion: Polymorphism of ABCB1 gene may be a good guide for predicting responsiveness for ondansetron.
Keywords: ATP-binding cassette subfamily B member 1 (ABCB1), Single nucleotide polymorphism, Genotyping, Ondansetron, P-glycoprotein. (JPMA 65: 963; 2015)
Introduction
A very common problem encountered in post-operative period is the complaint of nausea and vomiting despite the use of antiemetic treatment.1 This contributes to the distress faced by the patients. A serotonin receptor antagonist, ondansetron, is the commonly used drug for post-operative nausea and vomiting (PONV) due to its efficacy and safety profile.2 However, in spite of its use 35% patients still complain of PONV and it means that all the patients do not respond to the drug in the same way. So far, the reasons for this variability in antiemetic drug response are largely unknown. In addition to many risk factors being attributed to this nausea and vomiting, it is now realised that there may be a role of genetics responsible for this variability.3 Ondansetron is recognised by adenosine 5\'-triphosphate-binding cassette subfamily B member 1 (ABCB1) drug transporter in the blood brain barrier which in turn determines the concentration of drug in central nervous system (CNS). However, ABCB1 is highly polymorphic; the polymorphism of which may affect the expression and function of P-glycoprotein (P-gp) and thus can influence the drug disposition in the CNS, affecting efficacy and treatment outcomes. Much work has been done at nucleotide positions G2677T in exon 21 and C3435T in exon 27, but little attention has been given to C1236T in exon 12.4 Likewise, association polymorphisms in the ABCB1 gene and clinical responses have been investigated in different ethnic groups5-8 but there is no data available for our population. Determination of the prevalence of functionally important single nucleotide polymorphisms (SNPs) in this population is of great interest as one cannot simply translate the findings of pharmacogenetic studies in a population to another. Moreover, the findings of the studies which were performed in different clinical settings may not by definition hold true for highly emetogenic post-operative settings. These pharmacogenetical studies of antiemetic in anaesthesia are sparse. Studies are needed to translate the results of pharmacogenetical studies in this field into the clinical anaesthesia practice. Keeping the research done in cancer patients and other populations as base, we planned the current study to determine a possible impact of the C1236T polymorphism on treatment outcomes with prophylactic ondansetron in post-operative patients undergoing laparoscopic cholecystectomy under general anaesthesia.
We hypothesised that there may be a role of ABCB1 polymorphism in determining the response of post-operative patients to ondansetron and the polymorphism in this transporter gene may be playing a role in inter-individual variation.
Patients and Methods
The prospective, clinical trial was conducted in 2012-13, and comprised patients undergoing elective laparoscopic cholecystectomy. After approval was obtained from the Ethical Committee of the Centre for Research in Experimental and Applied Medicine (CREAM), Army Medical College, Rawalpindi, clinical data collection and sampling was done at Combined Military Hospital (CMH), Rawalpindi, and analytical procedures were carried out at the Institute of Biomedical and Genetic Engineering (IBGE), Islamabad. Written informed consent was obtained from all the subjects who were adults of either gender randomly selected through non-probability consecutive sampling belonging to different regions of Pakistan to provide representation from all areas.9 Patients aged between 18 and 65 years with an American Society of Anaesthesiologists (ASA) physical status of I or II scheduled to undergo elective laparoscopic cholecystectomy were enrolled. Patients with a history of any known hypersensitivity to 5-HT3receptor antagonist, hepatic or renal disease or who had used antiemetics within 24 hours before the study were excluded. Each subject was evaluated with detailed medical history which included the history of smoking, motion sickness and any past experience of PONV.
The vitals of the patient, including electrocardiography, non-invasive blood pressure (BP) and pulse oximetry, were monitored continuously once the patient was received in the operation theatre. Induction was carried out with 4-5 mg/kg thiopental, and endotracheal intubation was carried out with 0.6 mg/kg rocuronium. Besides, 1.5-2.0 vol% sevoflurane and nalbuphine 0.1mgkg-1 was used for maintenance of anaesthesia. During the procedure a bispectral index score (BIS) monitor was used continuously and depth of anaesthesia was maintained appropriately between 50 and 60. All the patients were given 4mg (0.1 mg/kg) ondansetron intravenously (IV) 30 minutes before the end of surgery. The total dose of nalbuphine consumption during anaesthesia was noted down. On arrival in the post-anaesthesia care unit, the pain score were recorded. In the post-operative period, all patients were observed for symptoms of nausea and vomiting. PONV in the first 2 hours and at 2-24 hours were recorded. Patients having any complaints of nausea or vomiting were allocated to the non-responders group. These patients were considered to have failed therapy and were given rescue antiemetic. Patients were allocated to the responders group if they did not complain of any nausea or vomiting post-operatively. A 5ml blood sample was taken from all the patients.
The standard organic methods of deoxyribonucleic acid (DNA) extraction were used to extract the genomic DNA from whole blood.10 The genotyping for C1236T was made by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). The genomic DNA was amplified using sense: 5\'GCCACaGTCTGCCCACTC3\' and anti-sense: 5\'CCCATaTCGAAAAGAAATTAAG3 primers for the region harbouring the C1236T SNP. The PCR was carried out in a final volume of 20µl containing 10X PCR buffer without Mg2+, 25 mM MgCl2, 2 mMdNTPs, 5U Taq polymerase, 10 µM forward and reverse primers and 40 og genomic DNA. The PCR products were subjected to digestion with restriction enzyme (Hae III). After digestion the homozygous individuals for major allele had three fragments of 272bp, 63bp and 35bp. The heterozygous containing both the major and minor allele yielded four fragments of 272bp, 98bp, 63bp and 35bp. The minor allele homozygous individuals produced two fragments of 272bp and 98bp fragments.
The data was analysed using SPSS 16. The frequencies of this SNP was assessed for deviation from Hardy-Weinberg equilibrium using Fisher\'s exact test. Frequency differences in genotype and incidence of PONV were compared by chi-squared test. The odds ratios (ORs) and 95% confidence intervals (CIs) used to determine the association of ABCB1 genotypic variants with the occurrence of PONV. P<0.05 was considered significant.
Results
Of the 426 patients, 70(16.4%) patients had CC genotype, 188(44%) had CT and 168(39.5%) had TT genotype (Table-1).
There were no significant differences in the characteristics and clinical details according to the genotypes (p>0.05). Among C1236T variants, the incidence of PONV during the first 2 hours after surgery was significantly lower in patients with the 1236TT genotype than other 1236 genotypes (TT vs CC + CT; p<0.001). The occurrence of PONV was significantly higher in patients with CC genotype at 2 hours than other 1236 genotypes (CC vs CT + TT; p<0.001) (Table 2).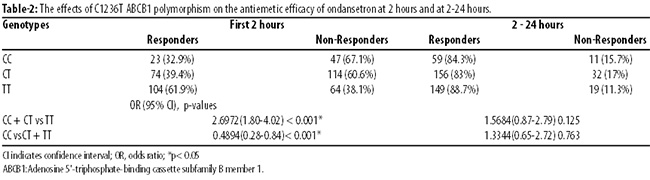
The response to ondansetron for PONV did not differ according to genotypes between 2 and 24 hours after surgery. The complaint of the patients with nausea and vomiting had decreased in this time period and it occurred only in 62(14.5%) patients.
Discussion
The transporter P-gp is encoded by a highly polymorphic gene ABCB1. One SNP that has been less widely studied in healthy populations as well as different clinical conditions is the mutation at position 1236 in exon 12 (C1236T). This polymorphism affects the expression and function of P-gp and thus efficacy of the drugs that are the substrates of P-gp.11 It is clear that polymorphism of ABCB1 analysis can provide important information to optimise the individualised therapeutic approach. However, there are inconclusive and contradictory results regarding the silent C1236T polymorphism in various populations. Some of the studies have supported the influence of genetic variability on the efficacy of antiemetics.12-14 Some studies have established an association for C1236T polymorphism and clinical outcome.5,12,15-17 while the others failed to do so.7,18
Differences in results among studies have been mostly attributed to phenotype definition, small sample size and overlap in substrate specificity between P-gp and other drug efflux transporters.19 We found that the response to ondansetron for PONV did differ significantly according to genotypes at 2 hours after surgery. We have observed that TT genotype at 1236 was associated with a better drug response at 2 hours in post-operative period. It is most possible that patients with 1236TT genotype have a more availability of ondansetron in the CNS and thus a better response to ondansetron, pointing to reduced activity of ABCB1transporter. We also observed that the patients with GG genotype had more complaints of PONV, thus pointing to the fact that it may be a predictor of poor response to this substrate of P-gp. Therefore, it is reasonable to hypothesise that genetic variance of this transporter could influence the efficacy of ondansetron. We found that the response to ondansetron for PONV did not differ significantly according to genotypes during between 2 hours and 24 hours after surgery.
It is believed that PONV is a multifactorial problem. Numerous risk factors have been described but only few seem to be unequivocally proven. The gender, the patient\'s history of PONV or motion sickness, non-smoking status, volatile anaesthetics, nitrous oxide, and opioids have shown to be independent predictors for PONV in various centres.20 This multifactorial genesis of PONV is currently regarded as a major cause of possible treatment failure. There are several risk factors affecting PONV. We had observed no significant differences in these risk factors in accordance with the genotypes. The other factors that affect PONV like the anaesthetic agent and type of surgery were also controlled in this study so as to minimally affect the results.
The response to ondansetron for PONV was significantly influenced by ABCB1 gene C1236T polymorphism in Pakistani population. ABCB1 genotypes may be a clinical predictor of responsiveness for ondansetron for PONV in this region of the world. Hence, genotyping of ABCB1 gene polymorphism (C12367T), including the TT, CT and CC genotypes, might be helpful in planning the individualised therapy based on the genetic makeup and the antiemetic therapy can be upgraded by identifying non-responders.
This study has served as one of the many efforts towards evaluating the impact of ABCB1genetic variability on the antiemetic efficacy in post-operative Pakistani patients. This polymorphism had never been studied in such a large group of patients so far. It has also served as a step ahead in the implementation of the concept of efficacious use of ondansetron by pointing out a possible role of ABCB1polymorphism C1236T for treatment outcomes. However, in order to implement genetic testing in routine clinical practice, further validation is required. It is suggested that the possible role of other polymorphisms of all the proteins that are contributing their part in the transport of drugs be also evaluated in order to combat the problem of drug resistance.
Conclusion
Polymorphism of ABCB1 gene may be a good guide for predicting responsiveness for ondansetron, and there is a role for genetics in the management of PONV with P-gp substrates.
References
1. Gan, T.J. Mechanisms underlying postoperative nausea and vomiting and neurotransmitter receptor antagonist-based pharmacotherapy. CNS Drugs. 2007;21:813-33.
2. Smith HS, Cox LR, Smith EJ. 5-HT3 receptor antagonists for the treatment of nausea/vomiting. Ann Palliat Med. 2012; 1:115-20.
3. Belle DJ, Singh H. Genetic factors in drug metabolism. Am. Fam. Physician. 2008; 77 : 1553-560.
4. Choi EM, Lee MG, Lee SH, Choi KW, Choi SH. Association of ABCB1 polymorphisms with the efficacy of ondansetron for postoperative nausea and vomiting. Anaesthesia. 2010; 65: 996-1000.
5. Zimprich F1, Sunder-Plassmann R, Stogmann E, Gleiss A, Dal-Bianco A, Zimprich A,et al. Association of an ABCB1 gene haplotype withpharmacoresistance in temporal lobe epilepsy. Neurology. 2004; 63:1087-9.
6. Kwan P, Baum L, Wong V, Ng PW, Lui CH, Sin NC, et al. Association between ABCB1 C3435T polymorphism and drug-resistant epilepsy in Han Chinese. EpilepsyBehav. 2007;11:112-7.
7. Lakhan R, Misra UK, Kalita J, Pradhan S, Gogtay NJ, Singh MK et al. No association of ABCB1 polymorphisms with drug-refractory epilepsy in a north Indian population.Epilepsy Behav. 2009; 14: 78-82.
8. Kim DW, Lee SK, Chu K, Jang IJ, Yu KS, Cho JY et al. Lack of associationbetween ABCB1, ABCG2, and ABCC2 genetic polymorphisms and multidrug resistance in partial epilepsy. Epilepsy Res. 2009; 84:86-90.
9. Löscher W, Delanty N. MDRl/ABCBl polymorphisms and multidrug resistance in epilepsy: in and out of fashion. Pharmacogenomics. 2009; 10:711-3.
10. Sambrook J, Fritich EF, Maniatis T. Molecular cloning: A Laboratory Manual, 2nd ed. New York, USA: Cold Spring Harbor Laboratory Press, 1989.
11. Marzolini C, Paus E, Buclin T, Kim RB. Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clin-ical relevance. ClinPharmacolTher. 2004; 75: 13-33.
12. Babaoglu MO, Bayar B, Aynacioglu AS, Kerb R, Abali H, Celik I, et al. Association of the ABCB1 3435C>T polymorphism with antiemetic efficacy of 5-hydroxytryptamine type 3 antagonists. ClinPharmacolTher. 2005;78:619-26.
13. Candiotti KA, Birnbach DJ, Lubarsky DA, Nhuch F, Kamat A, Koch WH, et al. The impact of pharmacogenomics on postoperative nausea and vomiting: do CYP2D6 allele copy number and polymorphisms affect the success or failure of ondansetron prophylaxis? Anesthesiology. 2005; 102:543-9.
14. Ho K, Gan T. Pharmacology, pharmacogentics, and clinical efficacy of 5HT3 receptor antagonists for postoperative nausea and vomiting. CurrOpinAnaesthesiol. 2006; 19: 606-11.
15. Hung CC, Tai JJ, Lin CJ, Lee MJ, Liou HH.Complex haplotypic effects of theABCB1 gene on epilepsy treatment response. Pharmacogenomics. 2005; 6: 411-7.
16. Seo T, Ishitsu T, Ueda N, Nakada N, Yurube K, Ueda K et al. ABCB1polymorphisms influence the response to antiepileptic drugs in Japanese epilepsy patients.Pharmacogenomics.2006 ;7:551-61.
17. Maleki M1, Sayyah M, Kamgarpour F, Karimipoor M, Arab A, Rajabi A, et al. Association between ABCB1-T1236C Polymorphism and Drug-Resistant Epilepsy in Iranian Female Patients.Iran Biomed J. 2010; 14: 89-96.
18. Kim YO, Kim MK, Woo YJ, Lee MC, Kim JH, Park KW, et al.Single nucleotide polymorphisms in the multidrug resistance 1 gene in Korean epileptics.Seizure. 2006; 15:67-72.
19. Löscher W, Delanty N. MDR1/ABCB1polymorphisms and multidrug resistance in epilepsy: in and out of fashion. Pharmacogenomics. 2009; 10: 711-3.
20. Gan TJ. Risk factors for postoperative nausea and vomiting.AnesthAnalg. 2006;102:1884-98.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




