Abdus Salam Khan ( Department of Basic Medical Sciences, Gomal University, D.I. Khan. )
Salman A. Malik ( Department of Biology, Quaid.e-Azam University, Islamabad. )
September 1996, Volume 46, Issue 9
Original Article
Abstract
Tumor necrosis factor and various haematological parameters were studied in 90 patients suffering from falciparum malaria. They were divided into three groups on the basis of haemoglobin level. The difference in haemoglobin level between group-I (Hb<7 gm/dl) and group-2 (Hb 7-10 gm/di), as well as group-I and group-3 was statistically significant. The geometric mean TNFa concentrations in group-i (193.9 pg/mi) and group-2 (132.2 pg/ml) were higher as compared to group-3; however, the difference was statistically non-significant. The TNF concentration in group-2 correlated negatively with haemoglobin level (r=-.43, p=.O5). As a whole, 21% patients had leukocytosis, 3% leukopenia, 46% increased ESR and 26% elevated levels of fibrin degradation products. The platelet count was done only in 4 patients with bleeding problems. Twenty-five healthy subjects were included in the study as controls. The difference between TNF and haemoglobin level in group-I and controls was statistically significant (p<.05, p<.001 each). The role of tumor necrosis factor in the production of these changes is discussed (JPMA 46:198, 1996).
Introduction
Patients suffering from malaria present witha numberof haematological changes of which anaemia is a major complication especially in those infected with plasmodium falciparum1. Other changes include eosinopenia, neutropenia. reactive lymphocytosis, thrombocytopenia and neutrophil leukocytosis. Falciparum malaria also causes accelerated turnover of coagulation cascade. In severe disease, there is increased fibrinogen consumption, but in most cases plasma concentrations are either normal or elevated2.
The pathogenesis of anaemia remains obscure. It results either from defective production or excessive destruction of red blood cells or a combination of both. In tropics, repeated attacks of malaria eventually lead to profound anaemia, especially, if there is associated chronic blood loss –from hookworm infection, malnutrition, pre3gnancy and persisting relapsing or recrudescent parasitaemia. 3
Oxford and Nairobi workers have studied tumor necrosis factor alpha (TNFa) in Kenyan children with severe. mild and without malaria. They confirmed, that TNFa levels correlate with parasite density and with the severity of malaria. What part this cytokine plays in the pathophysiology of malaria is not yet established, but the possible role of anti TNFa antibodies in cerebral malaria is under investigation. It was reported in 1993 that monoclonal antibodies against TNFa reduce fever in children with cerebral malaria4.
The aim of this study was to study the haematological changes in patients with Falciparum malaria. correlate them with tumor necrosis factor and compare them with other studies.
Patients and Methods
Sixty-nine Pakistani adults (52 males and 17 females) and 21 children (16 males and 5 females), admitted in District Headquarter Hospital. D.I. Khan between August, 1994 and Fcbruaiy, 1995, with acute symptoms of malaria due to plasmodiurn falciparum were studied. Diagnosis of malaria was made on blood film. Parasite count was done by counting the number of a sexual plasmodium falciparum parasites against 200 leukocytes in a thick film. Twenty five healthy subjects (20 males and 5 females) were used as controls. Patients were divided into three groups on the basis of haemoglobin level, white blood cell count and FDP level. Group I patients had haemoglobin of less than or equal to 7 gm/dl (n=9). group 2 between 7-10 gm/dl (n=l6) and group 3 over 10.0 gm/dl (n=65). Blood samples were taken within 24 hours of admission, before the initiation of antimalarial treatment. Laboratory investigations like total Icukocvte count. hacmoglobin level, red blood cells count, erythrocyte sedimentation rate (ESR) were done by conventional method and fibrinogen degradation products (FDP) by latex agglutination method. Platelet count was done only in patients with bleeding problems due to plasmodium falciparuum malaria.
For the determination of tumor necrosis factor alpha (TNFa) venous blood samples were taken, sera separated and stored at -20°C until cytokine assay. Serum concentrations of TNFa were measured by the quantitative immunocnzvmometric, ‘sandwich’ technique. Microtitre plates were coated with tuonoclonal antibodies specific to TNFa. Standards with known amounts ofTNFa and samples were pippeted into the wells and any TNFa present was bound by the immobilized antibody. After washing away any unbound sample protein and enzyme linked polyclonal antibody specific for TNFx was added to the wells and allowed to bind the TNFa which was bound during the first incubation. Following a wash to remove any unbound antibody-enzyme reagent, a substrate solution was added as described by Kwiatkowski5 using an enzyme linked immunosorbant assay.
Results
Of 90 cases, 69 were adults (52 males and 17 females) and 21 children (16 males and 5 females). Their ages ranged from 1 month to 70 years.
Haemoglobin level in patients with falciparum malaria
In group-i there were 9 patients (10%). Their mean haemoglobin level was 5.9±0.5 gm/dl and geometric mean of TNFcz concentration 193.9 pg/mI. In group-2 there were 16 patients (19%) with mean haemoglobin oF 9.0+0.93 g/dl and a geometric mean TNF concentration of 132.2 pg/nil. In group-3, there were 65 (71%) patients with mean haemoglobin level of 12.5±1.09 gm/dl and geometric mean TNF level of 108.94 pg/ml. The relationship between haemoglobin level and tumor necrosis factor in different groups of falcipanum malaria is shown in Figure 1.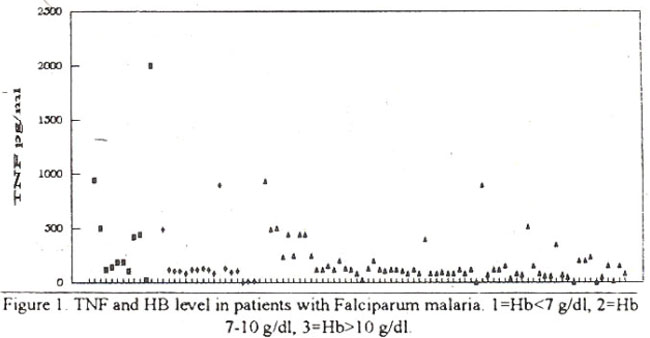
White blood cell count in patients with falciparum malaria with cell count in Group 1 (3 cases) was <4.Ox1O3/ul in Group 2 (68 cases) 4-11 Ox 10 /ul and in Group 3 (11 cases) it was>! 1.Ox1O3/ul (n=11).
Sixty-eight patients had normal leukocyte count, 3 had leukopenia and 19 (21%) had leukocvtosis. The relationship between WBC count and tumor necrosis factor is shown in Figure 2.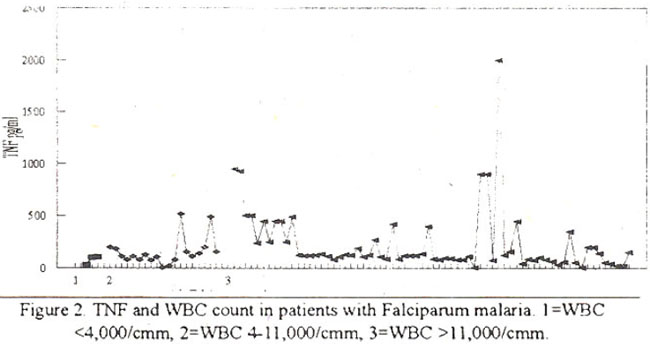
Fib rinogen degradation products (FDP level) in patients with falciparum malaria.
FDP level was less than 10 ug/dl in 74 cases belonging to group 1; between 10-40 ug/dl in 11 cases of group 2 and more than 40 jig/dl in 5 cases of group 3.
Seventy-four patients had normal FDP level (reference range 0-10 ug/dl, 11 patients between 10-40 ug/dl and 5 had levels above 40 ug/dl. The relationship between FDP level and tumor necrosis factor is shown in Figure 3.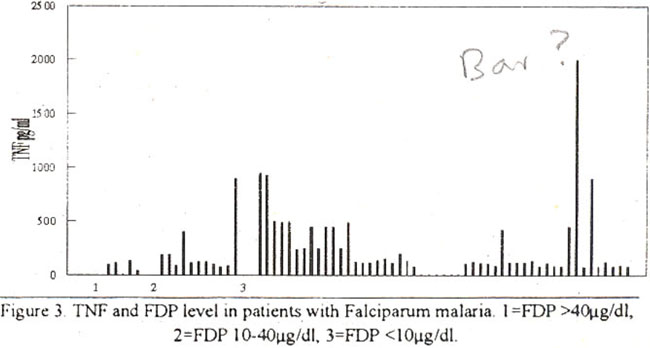
Platelet count in patients with falciparum malaria
Platelet count was done in 4 patients with bleeding problems, one had normal platelet count (150-400x109/i), while in 3 it was reduced (80-98x109/1). In 25 controls, the mean hacmoglobin level was 14.5±1.5. The geometric mean TNF level was 97.3 pg/mI (reference range 5-150 pg/mI) as shown in the Table.
In 3 patients with icukopenia TNF levels were with in reference range (5-150 pg/dl). Of 68 patients with normal white blood cell count, 25 (37%) had elevated TNF levels. Similarly in 19 patients with leukocytosis. 7 had increased TNF level. In 5 patients with markedly elevated FDP levels, TNF levels were normal. In 11 patients with moderately elevated FDP levels. 4 had elevated TNF levels. In 74 patients with normal FDP levels, 20 had increased TNF concentration.
Discussion
This study shows that falciparum malaria is associated with a number of haernatological changes like anaemia, leukocytosis, leukopenia, thrombocytopenia, high levels of fibrinogen degradation products and elevated erythrocyte sedimentation rate. It is further shown that the anaemia associated with falciparum malaria is related to the levels of tumor necrosis factor alpha (TNFa).
Anaemia is an inevitable consequence of ervthrocyte parasitization by the plasmodium falciparum as all infected cells are destroyed at shizogony. The survival of non-parasitized crythrocytes was found to be reduced for several weeks after clearance of parasitaemia in patients with falciparum malaria3. Many workers have described the correlation between high TNF level, cerebral malaria and hypoglycaernia, however, very few have reported the correlation between high TNF level and severe anaemia. Shaferetal found significant Iv elevated TNF levels in children with severe anaemia in Kinshasa (Zaire) 6. Gruss et al7 demonstrated that elevated TNFa and interleukin 6 (IL-6) levels are markers for complicated falciparum malaria. They also found higher incidence of severe anaemia in their patients as compared to our study. Eighteen percent of their patients had haemoglobin below 7.0 gm/dl.
There is now con.siderable evidence linking TNF with various aspects of malarial pathology10. The geometric mean TNF levels were much higher (193.9 pg/ml) in patients with severe anaemia (Hb 5.9±0.49) than in patients (108.94 pg/mi) with mild anaemia (Hb 12.5±1.09). Them was negative correlation between TNF levels and severe anaemia (r=.4, p.05). This observation shows that TNF may be involved in the production ofanaemia in patients with falciparum malaria. Elshoura8 reported several ultrastructural changes in non-parasitized eiythroblasts and erythrocytes in the blood of 28 Saudi patients who were anaemic and suffering from acute falciparum malaria. Ultra structural changes of these cells were suggested to be due to dyserthropoicsis and ineffective crthropoiesis which was attributed to an imbalance in metabolismas they were being overproduced in response to infection. Numerous haemoglobin like particles are liberated through erthrocyte plasma membrane, indicating severe hacmolysis which is one of the major factors producing anaemia during malarial infection.
Shofield and Hacket9 identified a dominant glvcolipid toxin of plasmodiurn falciparum. They showed that chemically it is glycosylphosphatidy linositol (GPI) and it induces tumor necrosis factor and inter leukin I production by macrophages. Thus GPI of plasmodium is a potent glycolipid toxin that may be responsible for novel pathogenic process, exerting pleiotropic effects on a variety of host cells9. The lower frequency of severe and moderate anaemia in our falciparum malaria patients may be due to the genetic diversity of the malarial parasite. The clinical immunity in our population may also play a relative protective role, as high infection rate with low prevalence of disease has already been demonstrated in rural Punjabi community in Pakistan10.
Leukocvtosis as well as leukopenia has been reported in falciparum malaria. Abdalla and co-workers showed that 29% of their patients with falciparum malaria and severe anaemia had leukocytosis11. Fourteen percent of our patients suffering from severe falciparum nrnlaria had leukocytosis, while 3% with mild malaria had leukopenia. Mild leukopenia has been described in uncomplicated malaria, buta neutrophilleukocytosis is an important abnormality in patients with severe falciparum malaria and is associated with a bad prognosis3. None of our patients even with marked Jeukocytosis died from severe falciparum malaria. Tumor necrosis factor may be responsible for leukocytosis which may be associated with a complicating Gram negative rod or other bacteraemia. However, 4 patients in our series with leukocytosis had TNF levels above 150 pg/ml3.
Serum fibrin degradation products are elevated in falciparum malaria. Tanabe and co-worker found that 57% of falciparurn malaria patients had elevated FDP levels. Twenty-nine percent of these patients had evidence of disseminated intravascular coagulation (DIC)12. Twenty-six percent patients in our series had elevated FDP levels. Eleven patients in the present study had cerebral malaria, but none of these had elevated FDP levels as reported by other workers13.
Thrombocytopenia is common in falciparum and vivax malarias. Its degree has some prognostic significance. Platelet survival is reduced to 2-4 days in severe falciparurn malaria. Increased numbers of large abnormal looking megakaiyocytes have been found in the marrow and the circulating platelets may also be enlarged suggesting dyspoetic thmmbopoiesis . Our study shows that although FDP levels are elevated in falciparum malaria, they have no pathological significance. The same observations have been made by Puknttayakamee and co-workers after studying coagulation profile in falciparum malaria14.
Our study shows that TNFa levels are elevated in patients with falciparurn malaria. Our data provide further confirmation of the association of haematological changes with falciparum malaria and that tumor necrosis factor may be directly or indirectly responsible for the production of anaemia and other pathological changes. The degree of pathology produced, can be the consequence of genetic variation in the host propensity to produce tumor necrosis factor. The decreased frequency of haematological abnormalities in our patients may be due to the presence of clinical inirnunjty in this community.
References
1. WeatheraH, D.J. and Abdalla, S.H. Anaemia of plasmodium falciparurn malaria. Br. Med. Bull. 1982;38: 147-151.
2. Pukrittayakarnee. S., White, N.J.. Clemens, R. et al. Activation of coagulation cascade in falciparum malaria. Trans. R. Soc. Trop. Med. Hyg., 1989;83 :762-66.
3. l3urce. C. Haematological changes in falciparum malaria Gilics, H.M. and Warrel, D.A eds. Essential malariology 3rd cd.. London, Edward Arnold, 1993. pp 53-54.
4. Nyakundi. J.N., Warn. P.. Newton, C. et al. Set-urn tumor necrosis factor in children suffering from plasmodium falcipanim infection in Kilifi District Kenya. Trans. R. Soc. Trop. Med. Hyg., 1994;88:667-70.
5. Kwiatkowski, D., Hill, A. VS. Sambou, D. TNF concentration and disease severity in fatal cerebral, non-fatal cerebral and uncomplicated Plasmodium falciparurn malaria. Lancet, 1990;336:1201-4.
6. ShalTer. N. Grau, G.E., Hedbcrg. K. et al. Tumor necrosis factor in severe malaria. J. Infect ,Dis., 1991;163:96-101.
7. Gruss, H J , 1)ietrich. M., Kern, P et al. Elevated tumor necrosis factor alpha and inten-leukin serum levels as markers for complicated falciparum malaria. Am. J. Med., 1989;87:139-43.
8. Elshoura, SM. The falciparum malaria in naturally infected human subjects. Ultra-structural alterations of non-parasitized red blood cells during anaemia.Appl Parasitol. 1993;34:173-79.
9. Shofield. I., and Hackctt, F. Signal transduction in host cells by aGlycosylphosphatidylinositol toxin of malaria parasites. 1. Exp. Med., 1993; 177:145-153.
10. Khaliq, A.A., Fox, E. Strickland, G.T. et al. Malaria in a Punjabi community. high infection rate with low prevalence of disease. Pak. J. Med. Res., 1985;24:67-74.
11. Abdalla, S.H., Wickramasinge, SN. and Weatherall. D.J The Deoxyuridine suppression test in severe anaemia following falciparum malaria. Trans. R. Soc. Tcop. Med. Hyg.. 1984;78:60-63.
12. Tanabe, K. and Shimada, K. Incidence of DIC complications in Japanese patients with malaria. Kausenshogaku-Zasshi, 1990;64: 1019.23.
13. Jaroonvesama, N. Intravascular coagulation in falciparum malaria.Lancet, 1972;n:221-23.
14. Pukrittayakamee. S., White, N.J., Clemens, R. et al. Activation of coagulation cascade in falciparum malaria. Trans. R. Soc. Trop. Med. Hug., 1989;83:762-66.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




